Chandra, Cosmos and Chemistry
The year 2011 has been proclaimed (by the United Nations and a host of other proclaimers) to be the International Year of Chemistry, a worldwide celebration of the achievements of chemistry and its contributions to the well-being of humankind.
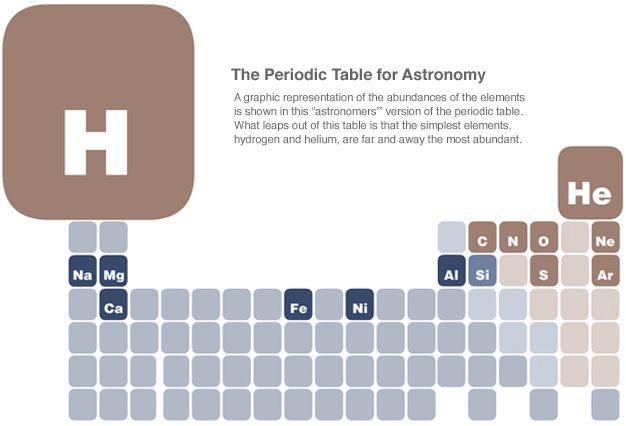
Chemistry, the study of the intricate dances and bondings of low-energy electrons to form the molecules of the various elements that make up the world we live in, may seem far removed from the thermonuclear heat in the interiors of stars and the awesome power of supernovas. Yet, there is a fundamental connection between them.
The formation of the elements began about 14 billion years ago, in the early minutes of the Big Bang. After about 20 minutes, the ordinary matter in the Universe was a mixture of 75% protons or hydrogen nuclei, 25% helium nuclei, and free-ranging electrons, which would start to be bound into atoms a few hundred thousand years later. In essence, the history of the formation of the elements can be divided into two main phases: one that ended after the first 20 minutes, and the other that has been ongoing since the formation of the first stars 13+ billion years ago.
-Wallace Tucker
Please note this is a moderated blog. No pornography, spam, profanity or discriminatory remarks are allowed. No personal attacks are allowed. Users should stay on topic to keep it relevant for the readers.
Read the privacy statement
